
Students will learn about the charges of each of the parts, protons, neutrons, electrons. They will learn about the basic structure of the atom. It is an ideal tool for differentiating instruction as it can be used as a station activity or as a practice for home to learn atomic structure basics. It is designed for students of physical science and chemistry. a) Fill in the blanks below to show your results: Neutral atoms have the same number of protons and. This practice sheet allows students to explore the atomic structure of six neutral, atomic elements. The nucleus of an atom is made up of protons, neutrons, and electrons. The smallest possible particle of an element is a (an) Atom. Play with the simulation to discover which particles affect the charge of an atom or ion. protons is equal to the number of electrons. In the reading, students will discover how the atom is the smallest unit of matter. a) An atom with 3 protons and 4 neutrons: Lithium b) An atom with 2 protons and 4 neutrons: Heleum c) An atom with 4 protons and 4 neutrons: Beryllium 5. Due to the uneven charges, they are considered ions and have an overall negative or positive charge. This station will provide students with a one page reading about atomic structure. If an atom looses or gains an electron, it becomes an ion. Because of the even charges, atoms are electrically neutral. Identify the three different types of elementary particles inside an atom, their electrical properties, and. Inside of each atom are several smaller bits of matter called particles. 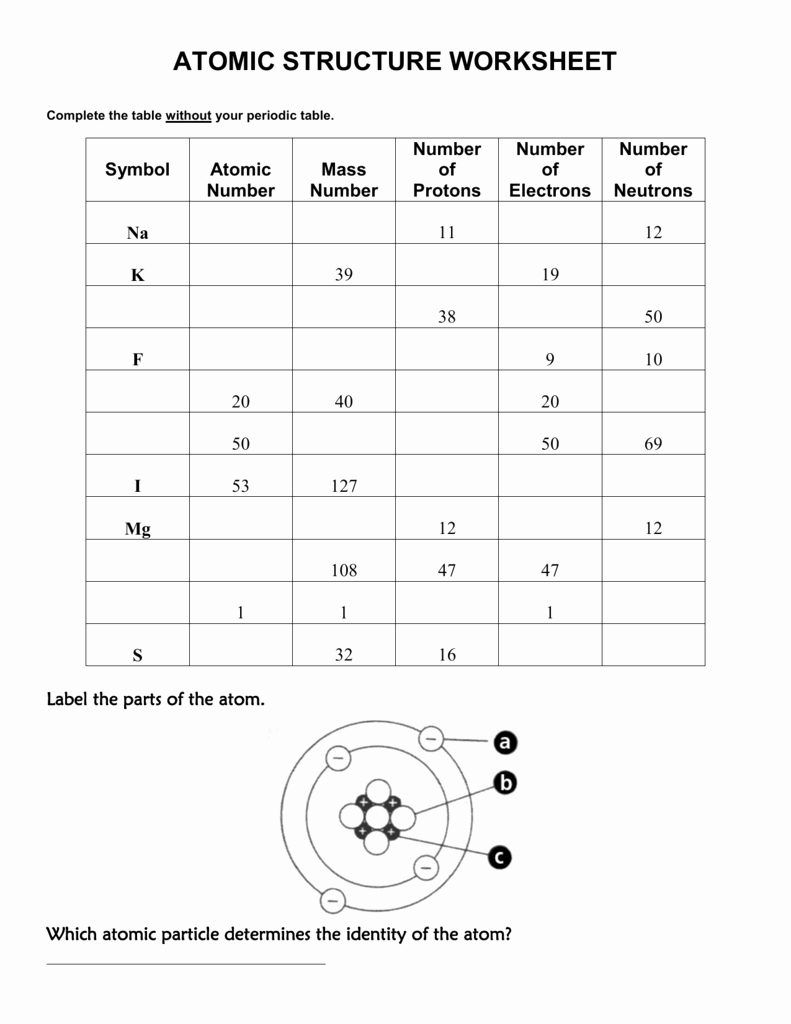
Because of their opposite charges electrons and protons are attracted to one another holding the nucleus together somewhat. Shown here is a simplified representation of an atom: the smallest division of matter that may be isolated through physical or chemical methods. In fact it has one-thousandths an atomic unit. Protons and neutrons have what is considered one atomic mass unit, but the electron has a lot less mass. Proton a subatomic particle inside the nucleus of an atom with a positive charge. Neutron it is a subatomic particle within the nucleus of an atom that has a neutral charge. Nucleus the dense center of an atom containing neutrons and protons. Honors chemistry summer assignment atomic structure worksheet. The mass of an electron is so small that it is generally not even considered. 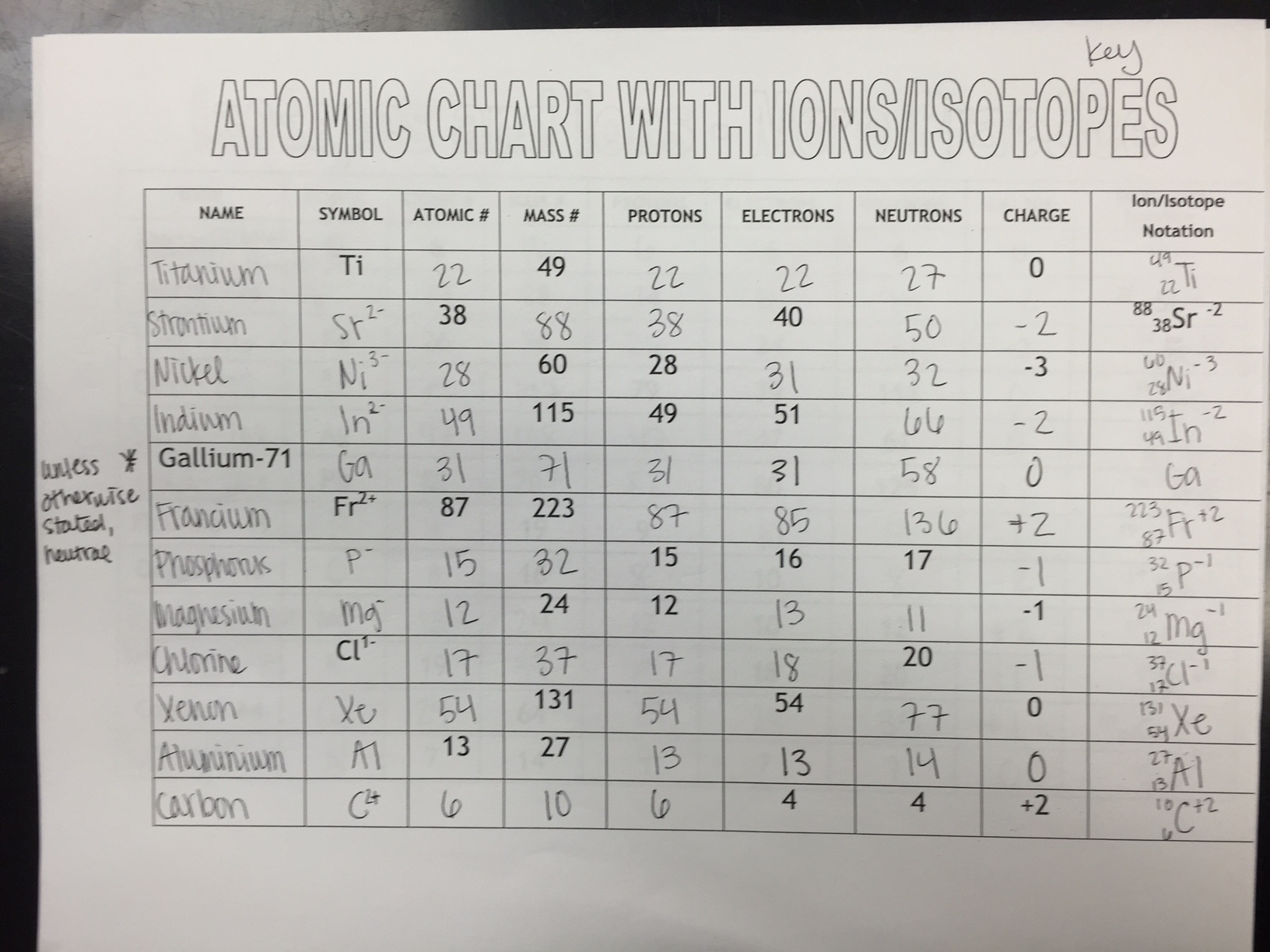
Here are the parts of an Atom: Electron it is a subatomic particle with a negative electrical charge. It has a dense nucleus with a cloud of negatively charged electrons surrounding it. I use this lesson to introduce the fifth graders in my school to the basics of atomic structure. Q 1 0 0 1 0 0 cm 1 1 1 rg 1 0 0 RG 0 w BT 0 0 0 rg /HelvBld 18 Tf 1 0 0 1 163.9217 745.3876 Tm (Atoms & Molecule Worksheet) Tj ET Qġ46 0 obj/M(D:20160204222139-08'00')/AP/Rect/NM(TWLITIQJJIXCRECL)/RC( Atoms & Molecule Worksheet Answer Sheet)/Contents >ġ49 0 obj>/Matrix/Length 241> Periodic Table Puns - Answer Key (pdf) Tips for Elements (pdf) Back to top Atoms Family (Lesson created by Kathleen Crawford, 1994) Targeted Concepts: Atoms, elements, subatomic particles, atomic structure. The information the students must fill out includes the atomic symbol, including appropriate ionic components where needed, atomic number, atomic mass, numbers of protons, neutrons, electrons and the atomic name. Challenge Work )/Contents(Challenge Work )>ġ27 0 obj>/Matrix/Length 183> This download includes a worksheet for the students to complete, as well as a key for you.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
